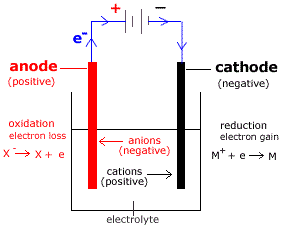
In a voltaic or galvanic cell, the anode is negatively charged while the cathode is positively charged. In an electrolytic cell, the anode is positively charged while the cathode is negatively charged. The following steps can help you figure it out. There is quite some confusion about the aspect of whether oxidizing agents accept or give away electrons. 
An electrode at which the chemical species loses electrons and the oxidation of the chemical species occurs is known as anode. The reactant that oxidizes the other reactants is called as the Oxidizing agent and reactant that reduces is called Reducing agent. Note: An electrode at which the chemical species gains electrons and the reduction of the chemical species occurs is known as cathode. Subtract the electrode potential of the anode from that of the cathode, and you get the electrode potential of the cell, or voltage: E o cell E o cathode - E o anode. Thus, the cathode is the reducing electrode. This is because the concentration of electrons is lower on the cathode.ĭuring the process of reduction, electrons are gained and cathode can gain electrons as it is positively charged. The cathode is a positively charged electrode. Thus, the anode is the oxidising electrode. This is because the concentration of electrons is higher on the anode.ĭuring the process of oxidation, electrons are released and anode has sufficient concentration of electrons to be released. The anode is a negatively charged electrode. Two metal electrodes called anode and cathode are immersed into the electrolytic solution. The electrolytic cell consists of a container filled with an electrolytic solution. These electrodes are dipped in a solution of an electrolyte One electrode is negatively charged and the other is positively charged. The cell consists of two electrodes, anode and cathode. Step by step answer: We know that a device which converts the electrical energy to chemical energy is known as an electrolytic cell. The anode has high concentration of electrons while the cathode has no electrons. Hint: The process in which the electrons are released is known as oxidation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
